Figure 1:
During the past few years the biotech industry has started to move away from protein A-based platform systems for MAb purifications. The high cost of protein A resins, their modest dynamic binding capacities (DBC), and their limited base stability place restrictions on a developmental engineer’s ability to reduce process costs and improve throughput for a given plant footprint.
Recent literature reports from Percivia (1) and Medarex (2) have described the use of improved cation exchange resins for MAb capture steps rather than more traditional protein A media. Scientists at Percivia were able to triple the loading capacity of the capture step with significant reduction in associated process costs, while process developers at Medarex, who incorporated precipitation with low pH conditioning for the feedstock, also reported loading capacities of up to 100 mg/mL resin (2).
These new cation exchange resins create a significant economic improvement parameter for developmental engineers.
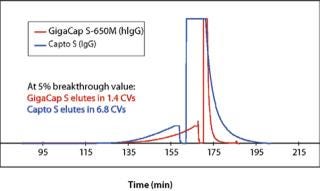
Percivia (1) selected Toyopearl GigaCap® S-650M resin (Capto™ S was also evaluated) for both its high MAb capacity and its ability to significantly reduce elution pool volumes because of its improved mass transfer kinetics.
Toyopearl GigaCap Ion-Exchange Resins
Tosoh Bioscience has introduced three new ion-exchange products, which together form the next generation of high capacity, polymeric ion-exchange resins for the purification of MAbs and other proteins.
The Toyopearl GigaCap series comprises three high capacity resins:
Toyopearl GigaCap CM-650M
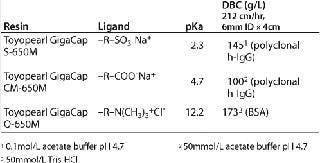
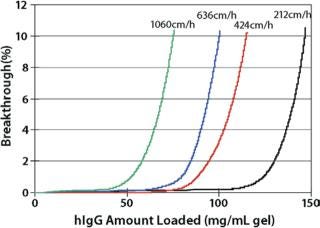
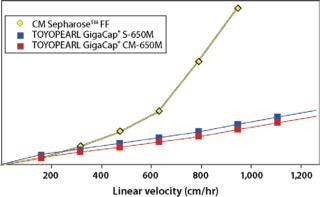
Each of the above resins exhibits high DBCs (Table 1) and each can be used at high linear flow velocities up to a pressure of 3 bar for improved process throughput. As reported by Lain et al. (1), for their PER.C6® MAb process, Toyopearl GigaCap S-650M maintains 90 mg/mL DBC even at LVs of 900cm/hr.
Table 1: Basic properties of Toyopearl GigaCap resins
Table 1: Basic properties of Toyopearl GigaCap resins ()
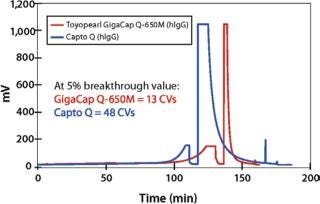
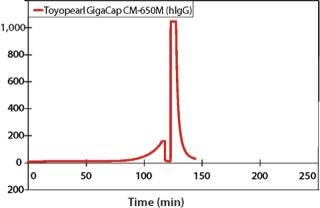
In addition, data in Figures 1, 2, and 3 show that proteins are eluted in significantly lower pool volume and can be collected as more concentrated target, when compared with other high capacity resins. (Please note that as of this writing, no comparable high capacity weak cation exchange resins were available to compare breakthrough curves to Toyopearl GigaCap CM-650M.) Also as confirmed by Lain et al. (1) pool volume was 50% less with Toyopearl GigaCap S-650M than Capto S for their MAb.
Figure 1: ()
Figure 2: ()
Figure 3: ()
Toyopearl GigaCap Base Beads
Unmodified Toyopearl HW-65C resin was used as the base bead for the new Toyopearl GigaCap resins. The average particle size of 75µm provides for enhanced efficiency and higher resolution than other larger particle size materials, while improved pressure-flow properties are obtained over smaller particle size materials.
Toyopearl GigaCap Ligand Attachment Chemistry
A polymer bead contains a distribution of small to large pores to which the ligand is normally attached relatively uniformly. The new Toyopearl GigaCap ligand attachment chemistry results in preferential placement of the functional groups in the larger more protein-accessible pores promoting both higher protein dynamic binding capacities and improved resin binding and desorption. The actual chemistry to obtain these high capacity ion-exchange resins is considered proprietary to Tosoh Corporation, Tokyo, Japan.
The basic properties of the Toyopearl GigaCap resins are listed in Table 1.
Toyopearl Gigacap S-650M (STRONG CATION EXCHANGE RESIN)
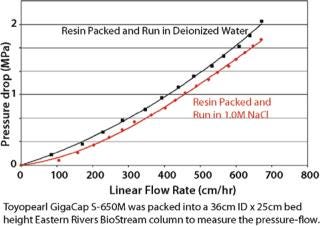
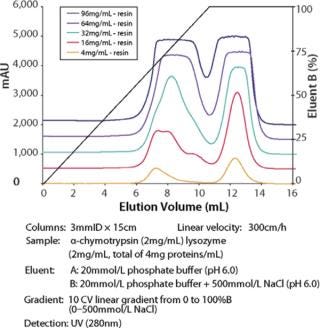
Toyopearl GigaCap S-650M resin was specifically developed for the purification of monoclonal antibodies. As other Toyopearl GigaCap resins, Toyopearl GigaCap S-650M resin has excellent kinetics (Figure 1) and maintains reasonably high capacities at higher linear velocities (Figure 4). The slightly larger particle size (50-100µm) has been optimized to give a unique combination of improved pressure-flow characteristics (Figure 5) with excellent resolution at high loads (Figure 6). In separate studies it was established that DBC-values for smaller proteins such as insulin and lysozyme were also impressive with typical values of 133 mg/mL and 167 mg/mL, respectively.
Figure 4: ()
Figure 5: ()
Figure 6: ()
Toyopearl GigaCap CM-650M (WEAK CATION EXCHANGE RESIN)
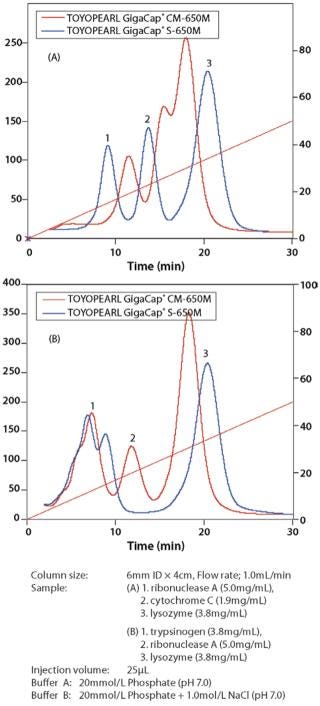
Toyopearl GigaCap CM-650M resin was also designed for the purification of monoclonal antibodies that require a different chromatographic selectivity than is available with Toyopearl Cap S-650M resin (Figure 7). Excellent kinetic properties and high capacity are maintained at high linear flow velocities. Because GigaCap CM-650M resin is based on the same particle size base-beads as the other members of the Toyopearl GigaCap series, very good pressure-flow properties are obtained for this resin as well (Figure 8).
Figure 7: ()
Figure 8: ()
Toyopearl GigaCap Q-650M (STRONG ANION EXCHANGE RESIN)
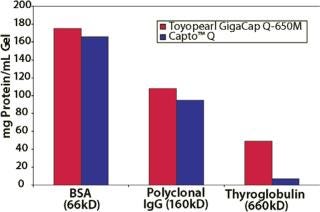
Toyopearl GigaCap Q-650M resin is the high capacity anion exchange resin in the Toyopearl GigaCap line of process chromatography resins. It was primarily designed for the capture and purification of proteins, although it can also be used for MAb polishing in negative or flow-through chromatography mode. Of particular note is the excellent capacity of Toyopearl GigaCap Q-650M for such large proteins as thyroglobulin (Figure 9).
Figure 9: ()
Conclusion
Over the past two years Tosoh Bioscience has introduced three Toyopearl GigaCap ion exchange resins (specifically designed for packed bed use), each offering high capacity at high linear velocities and a substantial reduction of target desorption volumes. These new products have the potential to improve process throughput even at manufacturing sites that have limited options to increased tankage.
Toyopearl and Toyopearl GigaCap are registered trademarks of the Tosoh Corporation.
PER.C6 is a registered trademark of Crucell Holland B.V. Corporation.
Sepharose and Capto are trademarks of GE Healthcare Biosciences.
Fractogel is a registered trademark of Merck KGaA.
REFERENCES
1.) Lain, B, M Cacciuttolo, and G. Zarbis-Papastoitsis. 2009. Development of a High-Capacity MAb Capture Step Based on Cation-Exchange Chromatography. BioProcess Int. 7:26-34.
2.) Arunakumari, A, J Wang, and G. Ferreira. Supplement to March 2009. Advances in Non-Protein A Purification Processes for Human Monoclonal Antibodies. BioPharm Int:22-26.