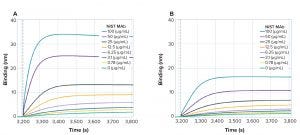
 A high-throughput method for relative screening of terminal sialic acid content was developed on the Octet platform to expedite cell line development. The method is based on the use of ForteBio’s sialic acid (GlyS) kit to bind sialic acid on glycoproteins. The GlyS kit can screen specifically the sialylation levels of secreted proteins in crude cell culture samples and does not require purified samples. Using this method, 96 crude cell culture samples can be screened for sialylation in 60 minutes or less. Here we used the GlyS kit to determine the relative sialylation level of representative glycoproteins, including erythropoietin (EPO), standard human monoclonal antibody (MAb) samples (NIST MAb), and fetuin glycoproteins, as well as crude biosimilar cell culture samples using an Octet HTX system. In a comparison study of the Octet GlyS kit method and high-performance liquid chromatography (HPLC), a linear correlation (R > 0.9) was observed between the GlyS kit binding signal (nm/g) obtained using an Octet HTX system and the total sialic acid content measured by HPLC. This GlyS kit method enables users to screen relative sialylation levels in crude samples in a high-throughput manner for the samples ranging between 1–25mol of total sialic acid per mol of protein, accelerating the process to monitor quality attributes early in cell line development for therapeutic proteins.
A high-throughput method for relative screening of terminal sialic acid content was developed on the Octet platform to expedite cell line development. The method is based on the use of ForteBio’s sialic acid (GlyS) kit to bind sialic acid on glycoproteins. The GlyS kit can screen specifically the sialylation levels of secreted proteins in crude cell culture samples and does not require purified samples. Using this method, 96 crude cell culture samples can be screened for sialylation in 60 minutes or less. Here we used the GlyS kit to determine the relative sialylation level of representative glycoproteins, including erythropoietin (EPO), standard human monoclonal antibody (MAb) samples (NIST MAb), and fetuin glycoproteins, as well as crude biosimilar cell culture samples using an Octet HTX system. In a comparison study of the Octet GlyS kit method and high-performance liquid chromatography (HPLC), a linear correlation (R > 0.9) was observed between the GlyS kit binding signal (nm/g) obtained using an Octet HTX system and the total sialic acid content measured by HPLC. This GlyS kit method enables users to screen relative sialylation levels in crude samples in a high-throughput manner for the samples ranging between 1–25mol of total sialic acid per mol of protein, accelerating the process to monitor quality attributes early in cell line development for therapeutic proteins.
Fill out the form below to read the complete technology review now.








