The vaccine manufacturing industry is continuously seeking more flexible and time-saving solutions for a quicker response to pandemic outbreaks (1, 2). For influenza, vaccine is traditionally produced in fertilized hen eggs and purified in fixed steel equipment. Here we describe the scale-up of a downstream purification process of cell-based influenza virus, from a 10 L cell culture to the purification from a 50 L culture using ReadyToProcess single-use technology. Single-use technology reduces the need for time-consuming device preparations, such as cleaning and cleaning validation, and minimizes the risk for cross-contamination, all essential parameters in traditional vaccine manufacturing.
Virus Purification
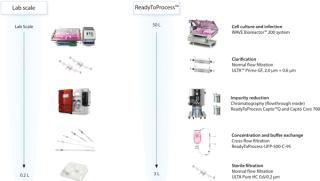
Influenza virus was propagated in Vero cells grown on microcarriers (3). Virus particles were subsequently purified using robust and scalable single-use filtration and chromatography techniques. In the laboratory-scale experiment, standard equipment was used for the downstream purification of influenza virus from a 10 L cell culture. The laboratory-scale purification process was subsequently scaled up 62.5-fold. In the scaled-up process, ReadyToProcess single-use equipment was used for downstream virus purification from a 50 L cell culture (Figure 1).
Figure 1: ()
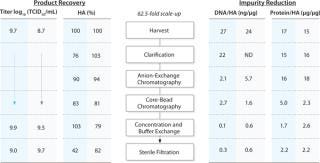
The outcome from the process steps of the influenza purification from both 10 L and 50 L production scales is shown in Figure 2. A more detailed description of the purification is given in Application note 29-04-35-49 (4).
Figure 2: ()
Comparison to Regulatory Specifications
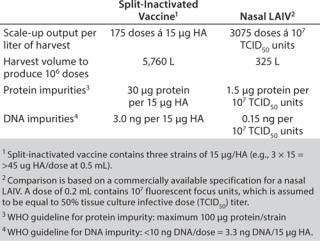
The output from the scaled-up production is compared with a commercially available specification for a nasal LAIV and a specification for egg-based, split-inactivated influenza vaccine from WHO (5, 6) (Table 1). Assuming a nasal route ofadministration and doses of 107 infectious particles per 0.2 mL dose, the amounts of host cell DNA and protein were shown to be below the acceptance levels defined by WHO. The outcome from the scaled-up production indicates that it is possible to obtain approximately 3000 doses/L harvest, corresponding to harvests of 325 L for 1 million doses (calculations based on specification for nasal LAIV) and 175 doses/L harvest corresponding to harvests of 5760 L for 1 million doses (calculations based on specification for split-inactivated vaccine).
Table 1:
Table 1:� 194; ()
Conclusions
In In this case study, the downstream purification of cell-based influenza virus from a 50 L cell culture using ReadyToProcess single-use technology demonstrates high productivity and efficient removal of host cell–derived impurities, with maintained virus infectivity. Similar process yield and purity was obtained from the scaled-up virus purification from a 50 L cell culture, as from the purification from a 10 L cell culture.
About the Author
Author Details
Peder Bergvall and Jakob Liderfelt are scientists, and Christine Sund Lundström is a senior research engineer at GE Healthcare Bio-Sciences AB, Björkgatan 30, 751 84 Uppsala, Sweden.
REFERENCES
1.) Pau, MG. 2001. The Human Cell Line PER.C6 Provides a New Manufacturing System for the Production of Influenza Vaccines. Vaccine 19:2716-2721.
2.) Kistner, O. 1998. Development of a Mammalian Cell (Vero) Derived Candidate Influenza Virus Vaccine. Vaccine 16:960-968.
3.) Application Note 29-0435 48, and AA Edition. 2013.Scale-Up of Adherent Vero Cells Grown on Cytodex Microcarriers Using ReadyToProcess Equipment, GE Healthcare, Uppsala.
4.) Application Note 29-0435-49, and AA Edition. 2013.Downstream Scale-Up Purification of Influenza Virus Using ReadyToProcess Equipment, GE Healthcare, Uppsala.
5.) 2005. Recommendations for the Production and Control of Influenza Vaccine (Inactivated). World Health Organization Tech. Rep. Ser 927:103.
6.) White Paper 29-0435-51, and AA Edition. 2013.Overview of a Scale-Up of a Cell Based Influenza Virus Production Process Using ReadyToProcess Equipment, GE Healthcare, Uppsala.