The complete characterization and analysis of biopharmaceuticals includes the application of size- exclusion chromatography (SEC) to measure protein aggregates and other size variants. Soluble protein aggregates in particular can contribute to immunogenicity. Accurate analysis and quantitation of biotherapeutic protein aggregates is therefore often required.
Current HPLC/silica-based SEC methods can be time- consuming and unreliable. Their uncertain results may be due to changes in retention time, peak shape, or spacing between peaks as well as irreproducibility between columns and changes in columns within a few runs. With the Waters ACQUITY UPLC® H-Class Bio System and below–2-µm ACQUITY UPLC BEH200 SEC column chemistry, SEC separations can be obtained reproducibly, reliably, and in shorter analysis time with minimal development. The ACQUITY UPLC H-Class Bio is a low- dispersion, high-pressure quaternary LC system that contains an inert flow path that, when combined with four-solvent mixing and Auto•Blend Plus™ technology, facilitates easy buffer preparation without pH adjustment.
The superior performance of this UPLC® SEC method relies on both the system attributes and the chemically stable BEH column. The combination of these components allows users to obtain more accurate and reproducible results over a larger number of samples than is observed with current SEC methodologies.
The ACQUITY UPLC BEH200 SEC particle has an effective diol coating that provides a stable particle with minimal secondary interactions. The packing material is more resistant to chemical and mechanical degradation over time. These attributes combine to provide an SEC column stable more than 600 injections and requiring lower buffer concentrations than traditional silica-based columns.
In a series of experiments, protein standards and monoclonal antibody biotherapeutics were analyzed with UPLC-based SEC. Repeated analysis of the same sample was performed at regular intervals over a two-day period. Reproducibility of the calibration was tested by analysis of proteins standards over the molecular weight range of 10,000–450,000 Da.
The elution volume for each protein standard was found to be within 0.2% RSD. The calibration curve points do not fall on a perfect straight line because the elution volume reflects both size and shape of protein standard. The consistency of the calibration curve is, however, indicative of both the column life and instrument control of flow rate and injection volume.
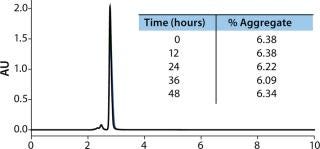
To test the reliability of quantitation, a humanized monoclonal antibody was analyzed. The sample shown (Figure 1) was found to have an average aggregate quantitation of 6.82% ± 0.3% of the monomeric species over the time period. The reliability of this analysis is demonstrated by the reproducibility of this measurement. The SEC separations demonstrate the accuracy and reproducibility of UPLC SEC technology, which, in turn, ensures accurate identification and aggregate determination.
Figure 1: ()
The ACQUITY UPLC H-Class Bio System used with an ACQUITY UPLC BEH200 SEC column provides reliable separations of proteins and their aggregates. As described, the analysis of both protein standards and monoclonal antibodies demonstrates the reliability of the calibration over a period of days. This reproducibility ensures accurate identification and quantitation of proteins and their aggregates, which can minimize analysis delays due to irreproducible results or incorrect peak identification. This, in turn, can increase laboratory throughput, thereby saving time and money.
About the Author
Author Details
Paula Hong is a senior applications chemist, Kenneth J. Fountain is manager of columns applications, and Damian Morrison is director of chemistry commercial operations at Waters Corporation, 34 Maple Street, Milford, MA 01757; 1-508-478-2000; [email protected], www.waters.com/hclassbio.