Figure 1:
New Brunswick Scientific has developed three new versions of Supervisory Control and Data Acquisition (SCADA) software programs. These SCADA programs allow fermentation or cell culture processes to be automated, monitored, and controlled. The three packages — BioCommand® Track and Trend, BioCommand Batch Control, and BioCommand Batch Control Plus — provide a variety of tools and the flexibility needed for any process requirement. Whether you require only basic trending and batch reporting, programming for process automation and optimization, or security and audit trails compatible with 21 CFR Part 11 regulations, there is a BioCommand package that will meet your needs.
In this article, the advantages of using a SCADA program will be explained and the differences between the three BioCommand packages will be identified. We will review the secure event logging and audit trail features of Batch Control Plus and provide an example of a control program created using BioCommand’s intuitive control editor.
SCADA vs. Local Control
Question: Why use a SCADA system at all, when most fermentors and bioreactors include built-in monitoring and control capability?
Answer: The trending and reporting tools, standard as part of all three BioCommand packages, are much more capable than those built into the NBS fermentor and bioreactor local system controllers. Integrated firmware is typically limited to basic graphical trending tools that simply allow a user to monitor each parameter over a finite period of time. Coupled with the system’s limited memory, these limitations may prove restrictive for longer processes. BioCommand software can perform much more complex tasks such as generating comparative graphs, graphing historical data, and comparing multiple parameters across concurrent processes. BioCommand uses PC memory onto which the software is installed. This feature allows for a comprehensive data logging and reporting tool that can provide easy access to important information from current or historical processes, thereby negating issues resulting from the local controller’s limited memory.
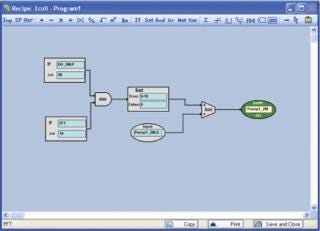
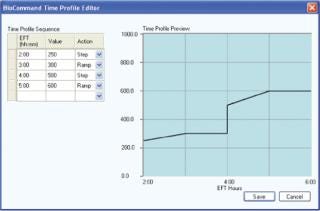
Although it is true that sophisticated, cascade-driven recipes can be programmed and executed directly from the local control systems on NBS fermentors and bioreactors, these recipes can be regulated only by parameter set-point values. BioCommand enables you to automate changes using logic-based control programming that can be driven by a multitude of factors including all parameter set-point values, elapsed fermentation time (EFT), and/or calculated values such as impeller tip speed (Figures 1 and 2). Additionally, BioCommand programming can be controlled from an off-site location via a remote connection thereby freeing the user from having to be in the lab at all.
Figure 1: ()
Figure 2: ()
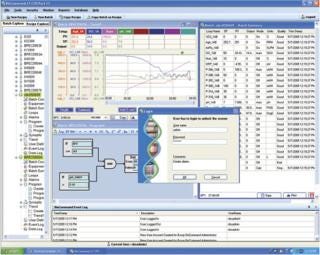
BioCommand Batch Control Plus, our premium package, is intended for users who are attempting to comply with 21 CFR Part 11 regulations. The software requires unique user names and passwords for all individuals performing any task during the process. A secure database logs all information associated with the process, including any changes made to the programming, as well as the required electronic signature/password accompanying the change. The reporting tool provides secure, printable (PDF) copies for all data associated with current and previous processes, which is the acceptable method of reporting according to most regulatory bodies (Figure 3).
Figure 3: ()
Laboratory and Production Packages
Question: Why are there three BioCommand packages?
Answer: Each package adds an extra level of functionality allowing users to choose a package that’s right for their application. All three BioCommand packages provide automated data logging; remote monitoring and set-point control capability; OPC compatibility (more fully explained below); the ability to monitor and supervise several fermentors/bioreactors from a single PC; recipe and alarm management; trend graphing to track multiple parameters from one or more process runs or compare historical and current values; and report logs detailing changes to process set-points and current values.
Additionally, Batch Control and Batch Control Plus packages add the following:
Advanced programming capability to optimize your process
Synoptic view providing a customizable pictorial representation of your process
Equipment lock-out feature preventing unauthorized users from changing set-point or alarm values directly through the fermentor/bioreactor.
For production facilities, Batch Control Plus adds the following:
Security — in fact three levels for operators, supervisors, and administrators, limiting allowable set-point and alarm changes through the software
Event log to automatically document batch events, capturing user name, date and time stamp, and a standard description of each change, such as “Batch Started”
Reports (such as “Event Log”), which cannot be altered, providing a secure, validatable record
OPC Compatible
Question: Can BioCommand connect to lab instruments other than NBS fermentors and bioreactors?
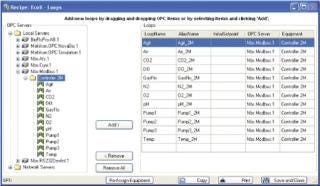
Answer: In a word, yes. Each new BioCommand software package runs on an OPC-compliant platform. Any OPC-compliant hardware or software package should seamlessly integrate with any of the New Brunswick software packages. BioCommand runs a search for all compatible signals during the initial phases of recipe construction. Once an instrument is recognized, the desired control loops can be selected and added to the recipe, making them available for use in the logic-based control programming as well as any of BioCommand’s data logging and tracking and trending tools (Figure 4).
Figure 4: ()
Summary
Whether you work in a lab or production facility, BioCommand will streamline data acquisition, monitoring, and control of your fermentation and cell culture runs. Advanced versions of BioCommand additionally help you optimize yields and meet FDA standards for verifiable documentation of the manufacturing process. For further information and sample screens, see www.nbsc.com/bim or contact New Brunswick Scientific at 1-732-287-1200 (toll-free in North America, 1-800-631-5417).