Change Is Difficult, But It Is Inevitable: The Value of Integrated Project Delivery to Biomanufacturing-Facility Construction
Agility, speed, and disruptiveness are now the “table stakes” in a world where the old rules governing biopharmaceutical manufacturing are being rewritten — in no small part by the COVID-19 pandemic and a new emphasis on lean project-delivery principles. All along the pharmaceutical value chain, companies, governments, and scientists worked at unprecedented speeds to mitigate SARS-CoV-2 outbreaks. Collaboration and constant communication among clients, trade partners, and vendors were critical at each step, from research and development (R&D) through commercial-scale production and distribution. Such experiences have revealed the value of integrated project delivery (IPD) as a holistic approach that supports facility design and construction to bring production of life-saving vaccines and therapies online without sacrificing cost, time, or quality.
But our company’s 2021 Horizons: Life Sciences report finds that despite such benefits, the biopharmaceutical industry seems to be making halting moves toward wide adoption of IPD (1). Despite important shortcomings with the traditional design–build (DB) and design–bid–build (DBB) project-delivery approaches, industry decision-makers still favor those methods over IPD. That raises an interesting question: What factors are behind the IPD reluctance, especially when the cost and scheduling benefits have become so evident?
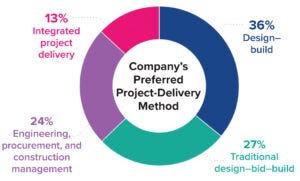
Figure 1: The 2021 CRB Horizons: Life Sciences survey asked participants, “What is your company’s preferred project-delivery method for large capital projects?”
The Power of Preference
In mid-2021, our company surveyed more than 500 life-science professionals to understand their impressions about the state of the pharmaceutical industry and where it seems to be going. In terms of facility-project delivery, the data show that even amid the COVID-19 pandemic, with disruptions happening across the industry, respondents ranked DB and DBB as their top project delivery models for large capital projects (Figure 1). DB came in as the preferred option with 36%, followed by DBB with 27%. Engineering, procurement, and construction management (EPCM) approaches came in slightly behind DBB (24%), and IPD ranked as the least preferred delivery method at 13%.
It is no secret that humans are creatures of habit; we often will use a method that has worked in the past — even despite its well-known disadvantages — over a newer, perhaps better option. DB and DBB have long provided benefits that make them both tried-and-true options for project delivery. Each approach comes with drawbacks, however, and our data indicate that the market increasingly is recognizing those limitations.
DB creates one central point of responsibility and has been known to ensure that facility projects are delivered on time. A pitfall is that DB can require much more project oversight than other methods. It also places a substantial amount of risk on project owners. Moreover, general contractors and/or subcontractors might add costs for embedded contingencies such as waste.
DBB often has the advantage of low upfront costs. Having a clearly defined and regulated bidding process drives down prices because of competition among bidders. Even though DBB can help companies minimize their initial capital spending, it creates risks for projects being over budget, sometimes with added costs and longer-than-expected project schedules. Low upfront price tags could become expensive in the long run, and a project might not finish on time. The DBB model also is prone to litigation if a project goes awry.
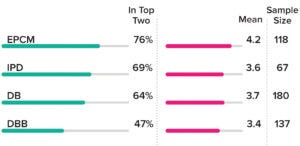
Figure 2: Breakdown of responses to a Horizons survey question asking, “On a scale of 1 (extremely dissatisfied) to 5 (extremely satisfied), how satisfied are you with your current project-delivery model?” Models included traditional design–build (DB); design–bid–build (DBB); engineering, procurement, and construction management (EPCM); and integrated project delivery (IPD) approaches. The “Top Two” column indicates how often respondents gave a model scores of “extremely satisfied (5)” or “somewhat satisfied (4).”
Participants’ responses highlight an interesting conflict: Even though DB and DBB ranked as the most-preferred project-delivery approaches, they received the lowest rankings in terms of respondent satisfaction with a current delivery model
(Figure 2).
EPCM scored highest in that regard. The model has become a mesh of approaches, lifting some of IPD’s collaborative principles. EPCM even could be considered as “IPD light,” a step toward full adoption of lean principles. Primary pharmaceutical manufacturers (producers of active pharmaceutical ingredients, APIs) tended to be the main proponents of IPD.
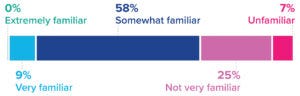
Figure 3: Responses to the question, “How familiar are you with ‘lean’ design and construction-project principles?”
Lack of Familiarity
IPD’s unpopularity among project-delivery methods comes as no surprise. The approach has been a viable option for only about a decade. That newcomer status perhaps explains the small numbers of respondents who indicated their familiarity with lean design and construction principles. Just 9% of survey participants reported being “very familiar” with lean design (Figure 3), and no respondents said that they were “extremely familiar” with it. Of note is that 20% of respondents at large pharmaceutical and biotechnology companies reported being “very familiar.”
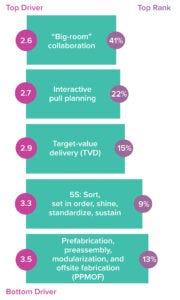
Figure 4: Participants ranked on a scale of 1 (most critical) to 5 (least critical) the following leanconstruction principles in terms of their importance to facility-project success. Bubbles on the left show mean scores. Bubbles on the right show how often a driver was selected as most critical.
However, IPD has made some inroads over the past decade, as indicated by the 58% bulge of participants with some understanding of lean design. Even more encouraging is that people familiar with the IPD approach showed a good handle on the importance of collaboration among lean principles (Figure 4). When asked to rank the criticality of lean principles to project success, respondents placed collaboration well ahead of other attributes, including interactive pull planning, target-value delivery, “5S” activities (sort, set in order, shine, standardize, and sustain), and prefabrication of key components. We expect that the benefits of “big-room” collaboration will play a significant role in helping to raise IPD’s reputation among biomanufacturers.
Holding Back from Change
The disconnect between how many survey respondents rely on DB/DBB and how few report feeling satisfied with the outcome raises an important question: What keeps organizations from transitioning — if not to IPD itself, then at least to EPCM? Our survey shows that companies are willing to change; an overwhelming 90% of respondents indicated that their organizations are open to adopting an alternative project-delivery method. But procurement seems to be the primary inhibitor to change (Figure 5).

Figure 5: Responses to the question, “If you are unsatisfied with your current project-delivery method, what are the top barriers to making a change?”
Both DB and DBB models appeal to those who want assurance on upfront price. But the healthcare, automotive, and industrial-manufacturing sectors (among others) have shifted away from DBB toward IPD methods, and surely, they too are driven by cost control. So other factors seem to be holding biopharmaceutical companies back.
In our experience, many such companies believe that the substantial amount of regulation in place on DBB projects helps to ensure fair, transparent bidding processes. In a highly regulated sector, a considerable amount of additional work is needed to prove that a new process and its associated materials comply with regulations. Thus, regulation is a good place to start when considering why other sectors have been faster to adopt IPD methods than the pharmaceutical industry has. But emphasizing regulation of upfront costs could exact a high price in turn. Strong, effective regulatory oversight is important, but too much focus on upfront pricing can preclude collaboration, shared risk, and simultaneous implementation — to cite just three of the leading benefits of IPD.
The reluctance of pharmaceutical companies to consider alternative delivery methods could stem from procurement leaders who are accustomed to DB and DBB. A company’s key performance indicators (KPIs) also could be structured in ways that discourage changes to delivery models.
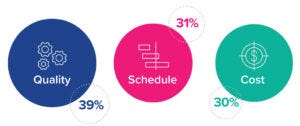
Figure 6: Responses to the question, “Which of these choices is your primary driver in the current market?”
Quality Is Key
Respondents ranked quality as the primary driver of successful project delivery. However, quality did not rank much higher than scheduling and cost, suggesting that all three factors are considered to be critical for successful delivery (Figure 6). The overarching goal of the lean project-delivery approach is to ensure that projects are delivered on budget, on time, and with superior quality. By definition, a truly lean approach is designed to deliver on all three attributes. Finding a close balance among quality, scheduling, and cost in the survey results reassures us of movement toward IPD in the sector, even if it isn’t happening as quickly as lean-approach proponents like.
Be Nimble, Quick, and Disruptive
Where is the biopharmaceutical industry on the path to general recognition of lean construction’s benefits and broad adoption of its principles? We can’t kid ourselves; if we imagine a 10-step path to universal acceptance of IPD, we’re probably at about step 3, and reaching full acceptance is unlikely to happen in this decade. But we have signs of movement, and industry clearly has recognized the shortcomings of traditional project-delivery models.
Certainly, we all want some good to have come out of the COVID-19 pandemic, which gave us tragedy, turmoil, and uncertainty. It is equally safe to say that the ability to pivot rapidly has never been held in higher regard in the biopharmaceutical industry because the public-health emergency forced a radical reconsideration of project timelines. The pandemic compelled a different way of doing facility design and delivery. Although the industry has not come around fully to implementing lean principles, it is reconsidering the benefits of traditional methods such as DB and DBB.
Because it holds promise for a “triple win” in ensuring quality, optimizing scheduling, and reducing costs, we expect that lean project delivery eventually will shift from being an option to an expectation. In a world constantly on the defense against health threats, that shift in thinking could have an enormous positive impact.
Reference
1 Horizons: Life Sciences, 2021. CRB: Kansas City, MO, 2021; https://go.crbgroup.com/horizons-life-sciences-report.
Further Reading
Seese C, Ballard C, Jarvis B. Plasma Facility Delivery: Three Reasons To Choose IPD. CRB: Kansas City, MO, 2023; https://www.crbgroup.com/insights/onesolution/plasma-facility-ipd.
Kearney D, Politi M. Improve Project Performance with PPMOF Strategies. CRB: Kansas City, MO, 2023; https://www.crbgroup.com/insights/construction/ppmof.
Swaney W, et al. Facing a Unique Challenge: Building an In-House Cell and Gene Therapy Manufacturing Facility During the Pandemic. BioProcess Int. 20(6) 2022: 10–18: https://bioprocessintl.com/manufacturing/facility-design-engineering/facing-a-unique-challenge-building-an-in-house-cell-and-gene-therapy-manufacturing-facility-during-the-pandemic.
Sinclair A, et al. Rapid Deployment of Manufacturing Options: An Analysis of Risks and Benefits. BioProcess Int. 19(5) 2021: 24–29; https://bioprocessintl.com/manufacturing/facility-design-engineering/rapid-deployment-of-manufacturing-options-an-analysis-of-risks-and-benefits-of-podular-construction.
Samavedam R. The Pandemic Changes Facility Strategy: A Focus on Speed to Market Triggers Innovation. BioProcess Int. 19(5) 2021: 47–48; https://bioprocessintl.com/manufacturing/facility-design-engineering/pandemic-changes-facility-strategy-a-focus-on-speed-to-market-triggers-innovation-in-cleanroom-space.
Mike Barrett is vice president of project delivery services, and Carl Rohs is senior project director for construction operations at CRB, 1255 Crescent Green, Suite 350, Cary, NC 27518; [email protected]; https://crbgroup.com.
You May Also Like




