Cell Therapies
Quantitative Risk Assessment of Bioaccumulation Attributable to Extractables and Leachables in Cellular Immunotherapy Biomanufacturing
Cell Therapies
Cost-Effective Process Development for Plasmid DNA Manufacture: Evaluation of Single-Use Technologies to Support <i>Escherichia coli</i> Culture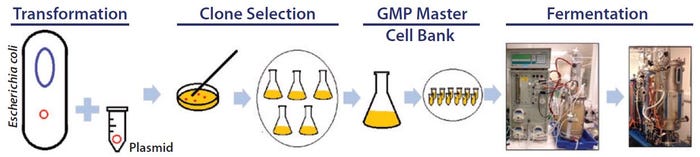
Cell Therapies
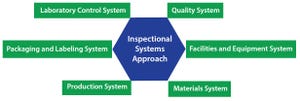
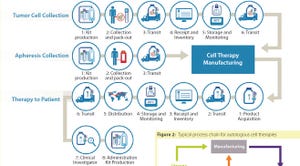
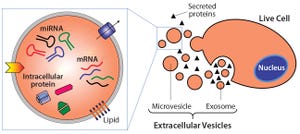
Planning for Commercial Scale of Cell Therapy and Regenerative Medicine Products, Part 2: Clinical Efficacy, Reimbursement, and Needle-to-Needle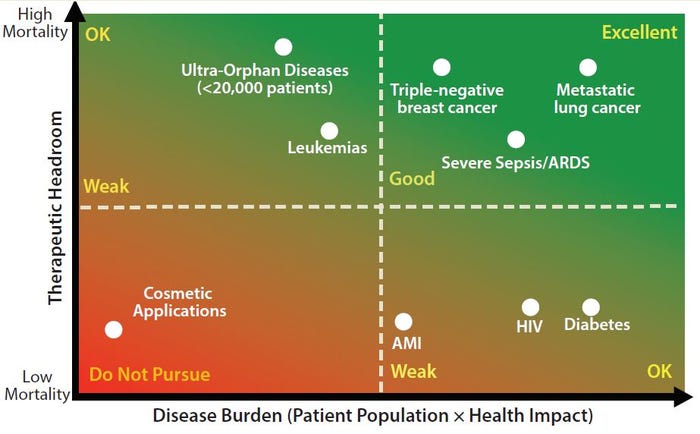
Cell Therapies
Planning for Commercial Scale of Cell Therapy and Regenerative Medicine Products, Part 2: Clinical Efficacy, Reimbursement, and Needle-to-Needle LogisticsPlanning for Commercial Scale of Cell Therapy and Regenerative Medicine Products, Part 2: Clinical Efficacy, Reimbursement, and Needle-to-Needle Logistics
Sponsored Content